|
Recent papers from this project:
Rius-Bonet J, Macip S, Closa D, Massip-Salcedo M. Intermittent
fasting as a dietary intervention with potential sexually dimorphic health
benefits. Nutr Rev. 2024 May 29:nuae062. doi: 10.1093/nutrit/nuae062.
Piletska E, Magumba K, Joseph L, Garcia Cruz A, Norman R, Singh
R, Tabasso AFS, Jones DJL, Macip S, Piletsky. Molecular imprinting as a
tool for determining molecular markers: a lung cancer case. S.RSC Adv.
2022 Jun 15;12(28):17747-17754. doi: 10.1039/d2ra01830f. eCollection
2022 Jun 14.
Poblocka M, Bassey AL, Smith VM,
Falcicchio M, Manso AS, Althubiti M, Sheng X, Kyle A, Barber R, Frigerio
M, Macip S. Targeted clearance of senescent cells using an antibody-drug
conjugate against a specific membrane marker. Sci Rep. 2021 Oct
13;11(1):20358. doi: 10.1038/s41598-021-99852-2.
Piletsky SS, Piletska E, Poblocka M, Macip S,
Jones DJL, Braga M, Cao TH, Singh R, Spivey AC, Aboagye EO, Piletsky SA.
Snapshot imprinting: rapid identification of cancer cell surface
proteins and epitopes using molecularly imprinted polymers. Nano Today
41, 101304, 2021.
Roach KM, Castells E, Dixon K, Mason S,
Elliott G, Marshall H, Poblocka MA, Macip S, Richardson M, Khalfaoui L,
Bradding P. Evaluation of Pirfenidone and Nintedanib in a Human Lung
Model of Fibrogenesis. Front Pharmacol. 2021 Oct 12;12:679388. doi:
10.3389/fphar.2021.679388. eCollection 2021.
Rada M, Qusairy Z, Massip-Salcedo M, Macip S.
Relevance of the Bruton Tyrosine Kinase as a Target for COVID-19
Therapy.Mol Cancer Res. 2020 Dec 16.
Kaur A, Macip S, Stover CM. An Appraisal on
the Value of Using Nutraceutical Based Senolytics and Senostatics in
Aging. Front Cell Dev Biol. 2020 Apr 3;8:218. doi:
10.3389/fcell.2020.00218. eCollection 2020.
Ekpenyong-Akiba AE, Poblocka M, Althubiti M,
Rada M, Jurk D, Germano S, Kocsis-Fodor G, Shi Y, Canales JJ, Macip S.
Amelioration of age-related brain function decline by Bruton's tyrosine
kinase inhibition. Aging Cell. 2019 Nov 17:e13079. doi:
10.1111/acel.13079.
Ekpenyong-Akiba AE, Canfarotta F, Abd B,
Poblocka M, Casulleras M, Castilla-Vallmanya L, Kocsis-Fodor G, Kelly
ME, Janus J, Althubiti M, Piletska E, Piletsky S, Macip S. Detecting and
targeting senescent cells using molecularly imprinted nanoparticles.
Nanoscale Horizons 4(3):757-768 01 May 2019
Tabasso AFS, Jones DJL, Jones GDD, Macip S.
Radiotherapy-Induced Senescence and its Effects on Responses to
Treatment. Clin Oncol (R Coll Radiol). 2019 May;31(5):283-289.
Rada M, Barlev N, Macip S. BTK: a two-faced
effector in cancer and tumour suppression. Cell Death Dis. 2018 Oct
18;9(11):1064. doi: 10.1038/s41419-018-1122-8.
Rada M, Barlev N, Macip S. BTK modulates p73
activity to induce apoptosis independently of p53. Cell Death Discov.
2018 Sep 11;5:30. doi: 10.1038/s41420-018-0097-7.
Rada M, Althubiti M, Ekpenyong-Akiba AE, Lee
KG, Lam KP, Fedorova O, Barlev NA, Macip S. BTK blocks the inhibitory
effects of MDM2 on p53 activity. Oncotarget. 2017 Nov
20;8(63):106639-106647. doi: 10.18632/oncotarget.22543.
Althubiti M, Macip S. Detection of Senescent
Cells by Extracellular Markers Using a Flow Cytometry-Based Approach.
Methods Mol Biol. 2017;1534:147-153.
Althubiti M, Rada M, Samuel J, Escorsa JM,
Najeeb H, Lee KG, Lam KP, Jones GD, Barlev NA, Macip S. BTK Modulates
p53 Activity to Enhance Apoptotic and Senescent Responses. Cancer Res.
2016 Sep 15;76(18):5405-14. doi: 10.1158/0008-5472.CAN-16-0690. Epub
2016 Jul 26.
Althubiti M, Lezina L, Carrera S, Jukes-Jones
R, Giblett SM, Antonov A, Barlev N, Saldanha GS, Pritchard C, Cain K and
Macip S. Characterization of novel markers of senescence and their
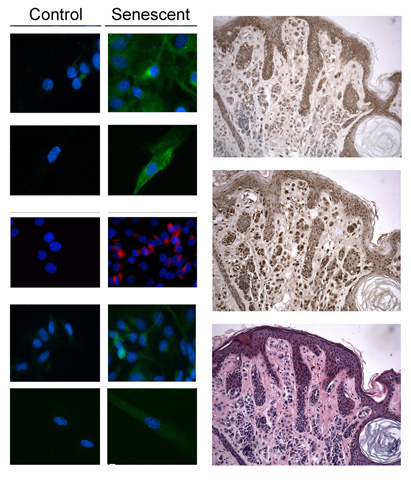
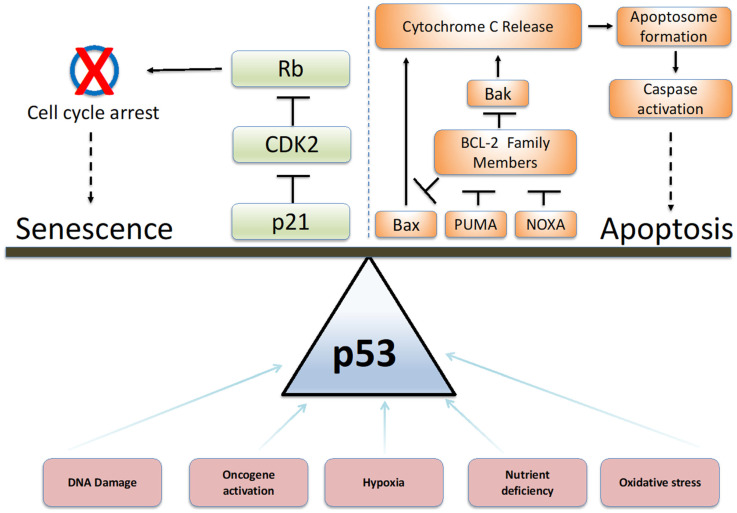
prognostic potential in cancer. Cell Death Dis. 2014 Nov 20;5:e1528.
doi: 10.1038/cddis.2014.489.
|